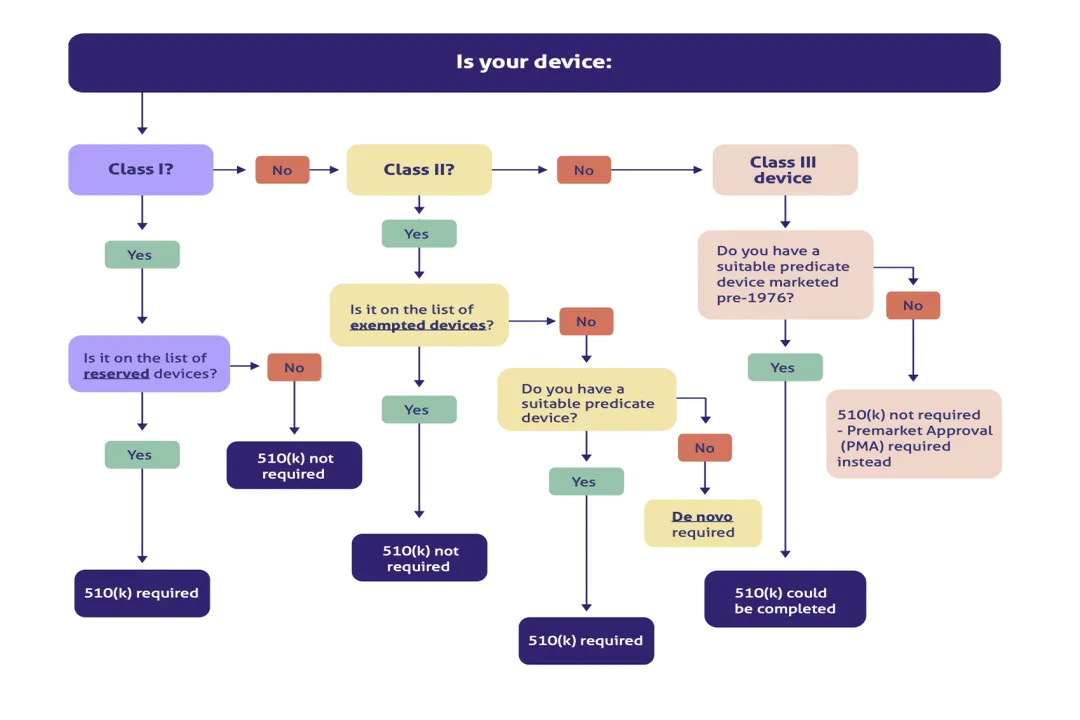
What criteria defines a product as a medical device by the FDA?
Answer
It is intended for diagnosis, treatment, or prevention without primary chemical action.
A product falls under the medical device category if it is used for diagnosing, curing, mitigating, treating, or preventing disease, or affecting body structure or function. Crucially, it must not achieve its primary purpose through chemical action, distinguishing it from pharmaceutical products, and it must comply with specific standards to avoid being considered adulterated under the law.
Related Questions
What does a Professional Use Only label primarily signify for a medical device?What criteria defines a product as a medical device by the FDA?How does the primary control of an Rx Only device differ from Professional Use Only?Why are Class II medical devices often restricted to professional use?What entity is responsible for vetting users of a professional device in a hospital?What is a requirement for providing electronic Instructions for Use (eIFUs)?Why are these devices sold through specialized medical distributors?What evidence is required to maintain accreditation for a facility using these tools?What is the purpose of the International Medical Device Regulators Forum?How does the Professional Use Only designation relate to risk classification?