What is a professional use only medical device?
Determining the specific classification and intended market for a medical device is a fundamental step in bringing that product to fruition, and sometimes a device is strictly designated for use by trained individuals rather than direct consumer purchase. A device labeled "Professional Use Only" signifies that its design, complexity, or inherent risk profile necessitates that its application, interpretation, or maintenance be performed exclusively by a licensed practitioner or someone operating within a specialized clinical or laboratory setting. [1][5] This is a critical distinction governed by regulatory bodies globally, designed to safeguard public health by ensuring that potentially complex or hazardous tools are handled only by those with the appropriate background and training. [6][7]
# Device Status Basis
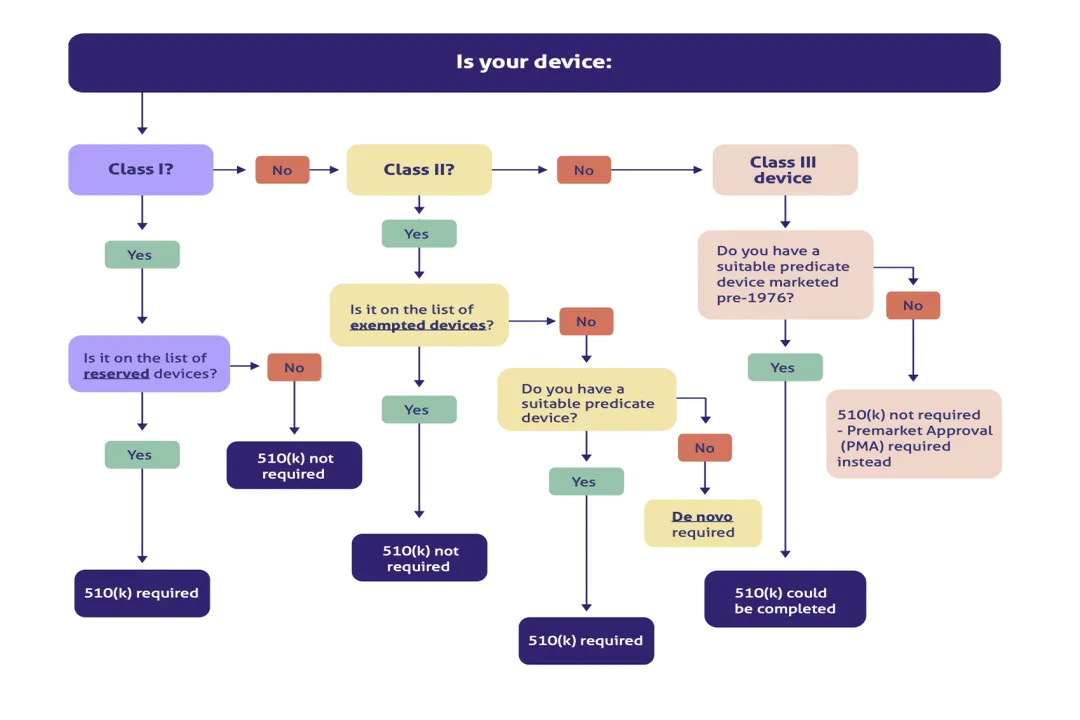
Before a product can earn any specific designation, it must first be classified as a medical device under the governing jurisdiction. In the United States, the Food and Drug Administration (FDA) determines if a product meets the definition of a medical device, which includes instruments, apparatuses, implants, in vitro reagents, or related articles intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, or intended to affect the structure or any function of the body, and which does not achieve its primary intended purposes through chemical action within or on the body, and which is not subject to being adulterated under the drug provisions of the Food, Drug, and Cosmetic Act. [1][6] If a product falls under this definition, its pathway to market hinges on its risk classification—usually Class I, II, or III. [6]
The designation of "Professional Use Only" is often tied to the device's intended use and labeling, which dictates who the manufacturer expects to be using the product. [1] For instance, a Class II device, which generally presents a moderate level of risk, might require special controls because of its complexity, leading the manufacturer to specify professional use to ensure those controls are met. [2][6] If a product's intended use is for diagnosis or treatment that necessitates the supervision of a licensed practitioner due to the potential for patient harm without that supervision, it will carry this restriction. [1]
# User Restriction Context
Regulatory agencies mandate strict control over distribution and labeling based on the intended user. In the European Union, for example, the classification rules under the Medical Device Regulation (MDR) dictate that devices intended only for use by a "healthcare professional" are restricted accordingly. [5] This restriction is a safety measure, ensuring that the inherent risks associated with the device are managed by trained personnel. [7]
When a manufacturer designates a product for professional use, they are effectively narrowing the scope of its accessibility. This means that the device should not be sold directly to the general public or the end-user patient. [3] The regulatory documentation, particularly the labeling, must clearly communicate this limitation to distributors and end-users alike. [1][5]
# Prescription Versus Professional
A common point of confusion arises when distinguishing between a "Prescription Use Only" (Rx Only) device and a "Professional Use Only" device, as both restrict direct consumer access. [2] While there is overlap, the core difference lies in the mechanism of control.
An Rx Only designation, often regulated under 21 CFR Part 801 in the US, generally means the device can only be legally dispensed pursuant to the prescription or, in the lawful or Indian Medical Device Order of a practitioner licensed by law to order such devices. [4] The control is centered on the prescription requirement for dispensing the product to the patient. [4]
Conversely, Professional Use Only focuses on the application or interpretation of the device. [2][3] A Professional Use Only device might not require a prescription for its purchase by a clinic, but its use is restricted to trained professionals. [2] A useful point of comparison involves certain diagnostic kits: an Rx Only test might still be administered by a professional, but the "Professional Use Only" label emphasizes that the professional is the only authorized operator, irrespective of whether a patient-specific prescription triggered the test in the first place. [2][3]
| Restriction Type | Primary Control Point | Typical User Environment | Dispensing Requirement |
|---|---|---|---|
| Prescription Use Only (Rx Only) | Legality of dispensing to the patient | Clinical or patient home setting (post-dispensing) | Requires a valid prescription [4] |
| Professional Use Only (POU) | Legality of operation or interpretation | Clinic, hospital, or laboratory | Purchase often restricted to licensed entities/professionals [3] |
A critical insight here revolves around institutional compliance. When an organization, like a hospital system, procures a Professional Use Only device, the responsibility shifts slightly from the individual practitioner's prescription authority to the institution's vetting process. The organization must ensure that only qualified personnel—those trained on that specific device—are accessing and operating it, as the supply chain is designed to bypass the general consumer market entirely. [3]
# Labeling and Information Display
The information presented on the device labeling and accompanying documentation is the primary mechanism regulators use to enforce the Professional Use Only status. [1] For devices intended only for professional use, the manufacturer must ensure that the labeling clearly states this limitation. [1]
In the EU context, the requirement for clear labeling is reinforced by provisions concerning Instructions for Use (IFU). For devices restricted to professional users, there is an increasing move toward extending the use of electronic IFUs (eIFUs). [5][9] While this offers environmental benefits and easier updates, manufacturers must be extremely careful that the delivery method (e.g., a dedicated website or QR code) remains accessible and reliable within the professional setting where the device is legally used. [9] A failure to provide immediate access to the IFU in a high-pressure professional environment could be interpreted as a labeling violation or even a safety issue. [5]
Specific content that must often be present on the labeling for POU devices includes:
- A clear statement indicating the device is restricted to use by licensed or trained professionals. [1]
- Warnings or precautions related to operator skill or necessary environmental controls. [1]
- Details on the required training background or certification necessary for safe operation. [5]
# Global Standards Alignment
The concept of restricting use to qualified personnel is not unique to individual national bodies like the FDA or the European Commission; it is a globally recognized principle for risk management. [7] The International Medical Device Regulators Forum (IMDRF) works toward harmonizing terminology and risk classification, which underpins these use restrictions. [7] A device deemed high-risk or complex in one major jurisdiction is highly likely to carry similar restrictions elsewhere, based on its technical characteristics rather than local consumer protection laws alone. [7]
This alignment means that multinational healthcare providers can generally expect consistency. If a piece of imaging equipment is designated POU in the US due to its complex calibration needs, it will likely require similar controls, such as dedicated service engineers or certified radiology technicians, in Canada, Japan, or Europe. [7]
# Operational Considerations for Professionals
The "Professional Use Only" stamp translates directly into operational requirements that differ significantly from consumer-grade products. When a professional buys such a device, they are not just buying equipment; they are assuming accountability for the user qualifications. [1]
# Training Burden Analysis
The expectation for professional use implies that the end-user possesses a baseline level of scientific knowledge, anatomical understanding, or technical skill relevant to the device's function. [6] For instance, a sophisticated in vitro diagnostic device meant for professional use requires laboratory staff trained in sample handling, assay procedures, and quality control checks. [1] If the manufacturer issues a warning that the device must only be serviced by factory-authorized personnel, that too falls under the professional use umbrella, as it dictates who can maintain the device's specified performance characteristics. [1]
This leads to an important consideration often overlooked by smaller clinics: documentation of competency. While the FDA focuses on the manufacturer's labeling, an accredited facility often requires internal records proving that the technician operating the POU device has completed the manufacturer’s specific training module. A simple list of employees is often insufficient; evidence of successful competency checks following training is necessary to maintain accreditation standards, especially when dealing with devices that directly impact patient diagnosis. [6]
# Supply Chain Integrity
The enforcement of POU status relies heavily on maintaining an unbroken chain of custody involving only authorized entities. [3] Sellers of these devices, particularly those active in large online marketplaces, must be vigilant about preventing diversion into consumer channels. [3] If a seller knowingly allows the sale of a POU device to an untrained individual or an entity that cannot verify professional status, they risk regulatory action. [2] This is why, unlike many consumer electronics, these devices are generally sold through specialized medical distributors who verify the purchasing entity's licensing or registration number. [3]
The regulatory environment acknowledges that if a device is sold outside this controlled loop, the manufacturer's ability to ensure safety and effectiveness through appropriate use protocols is compromised, thus invalidating the original clearance or approval. [1][6]
# Device Classification Overlap
It is worth noting that the Professional Use Only designation is often layered on top of a device's base risk classification rather than being a classification itself. [2] A device can be:
- Class I (Low Risk): Generally exempt from premarket notification, but labeling can still restrict it to Professional Use Only if the intended use requires professional oversight (e.g., specialized dental tools).
- Class II (Moderate Risk): Most likely to carry POU restrictions due to special controls required for safety and effectiveness, such as complex monitoring equipment. [6]
- Class III (High Risk): Almost certainly restricted, often requiring premarket approval (PMA) and involving significant clinical data that supports professional use exclusively, such as implantable devices. [6]
In summary, when a device carries the "Professional Use Only" restriction, it is a direct regulatory instruction that moves the responsibility for safe operation and interpretation from the patient to the qualified professional, backed by explicit labeling requirements and a restricted distribution channel. [1][5]
Related Questions
#Citations
How to Determine if Your Product is a Medical Device - FDA
Rx Only vs. Professional Use Only - Class II Private Label Medical ...
How to List Product's as a Professional Medical Device Use only?
21 CFR 801.1 -- Medical devices; name and place of ... - eCFR
[PDF] MDCG 2021-24 Guidance on classification of medical devices
The 3 FDA medical device classes: differences and examples ...
[PDF] Principles of Labeling for Medical Devices and IVD Medical Devices
41 CFR § 102-40.190 - How do we handle medical devices?
[PDF] Electronic Instructions for Use for all professional use Medical Devices:

