What is an example of T cell therapy?

T cell therapy represents a cutting-edge area of cancer treatment where the body's own immune cells are reprogrammed to seek out and destroy malignant cells. The most frequently discussed and FDA-approved example of this type of treatment is Chimeric Antigen Receptor (CAR) T-cell therapy. [2][3] This approach takes the fundamental power of the T lymphocyte—a white blood cell crucial for immune surveillance—and enhances it with synthetic biological machinery, essentially giving the T-cell a highly specific homing beacon for cancer. [3]
# Cell Transfer Concept
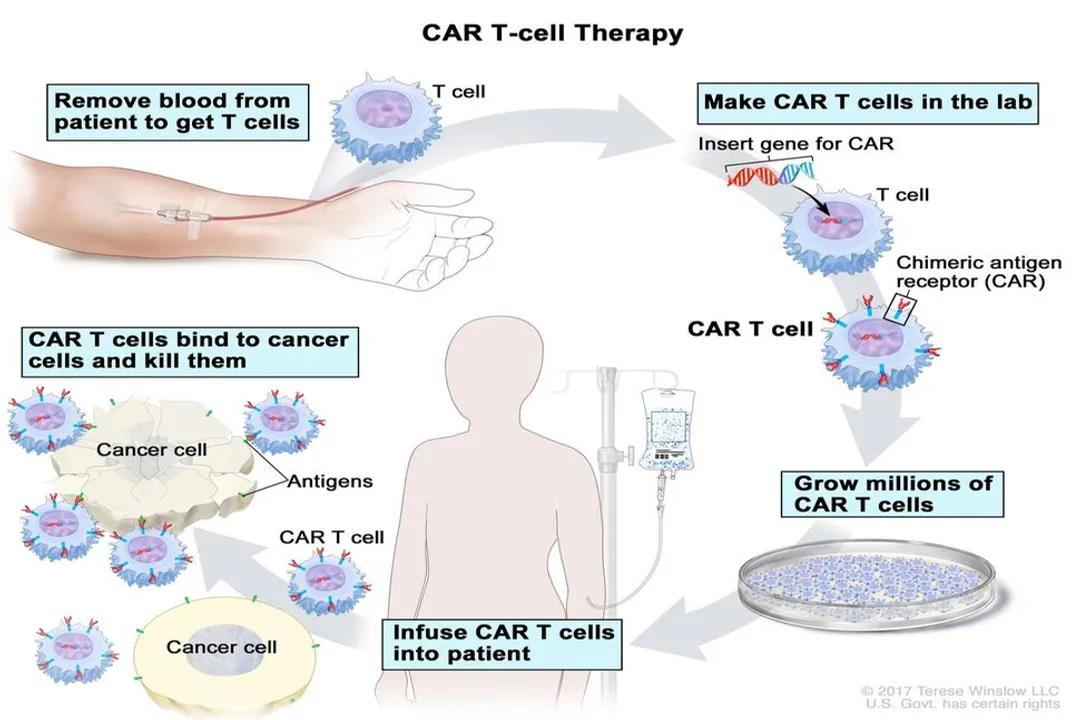
T cell therapy, in its broadest sense, often falls under the umbrella of adoptive cell transfer. [1] This concept centers on harvesting a patient's immune cells, modifying them outside the body to boost their cancer-fighting ability, expanding their numbers dramatically, and then reintroducing them intravenously back into the patient. [1] While CAR T-cell therapy is the most prominent example, the field also encompasses other techniques, such as Tumor-Infiltrating Lymphocyte (TIL) therapy, where T-cells naturally found within a tumor are selected and expanded. [1] The goal across these methods is to restore or amplify the body’s natural ability to recognize and eliminate cancer, which cancer cells often manage to suppress or evade through various defense mechanisms. [4]
# CAR T Mechanics
The modification that defines CAR T-cell therapy is the addition of the Chimeric Antigen Receptor (CAR). [2][3] This receptor is a custom-built protein grafted onto the T-cell surface. [3] The "chimeric" nature means it combines elements from different sources to achieve a specific function. It typically has an external binding domain that recognizes a specific protein—an antigen—on the surface of cancer cells, much like a lock-and-key mechanism. [2] Crucially, the intracellular portion of the CAR contains signaling domains that activate the T-cell upon binding, prompting it to attack and kill the target cell. [3]
For many approved treatments, the target antigen is the CD19 protein, which is prominently displayed on the surface of most B-cell cancers, including certain types of leukemia and aggressive lymphomas. [2][4][9] By equipping the T-cells with a CAR specific to CD19, these modified cells can specifically hunt down and eliminate any B-cell expressing that marker, whether the cancer originated in the bone marrow or has spread elsewhere. [3][9] This precision distinguishes it sharply from treatments like traditional chemotherapy, which lacks this specificity. [4]
# Treatment Steps
The creation of a personalized CAR T-cell product follows a distinct, multi-stage protocol. [1][4]
- Collection: The process begins with collecting the patient’s T-cells. This is usually accomplished via a procedure called leukapheresis, which is similar to donating platelets, where blood is drawn, the white blood cells (including T-cells) are separated, and the remaining blood components are returned to the patient. [3]
- Engineering: The collected T-cells are sent to a specialized manufacturing facility. [1] Here, a vector, often derived from a deactivated virus, is used to deliver the genetic blueprint for the specific CAR into the T-cells. [3] This genetic alteration permanently equips the T-cells with the ability to express the new receptor. [2][4]
- Expansion: Following successful engineering, the modified cells must be multiplied into a therapeutic dose, which often involves billions of cells. [4] This in vitro expansion phase allows the small initial sample to grow into enough cells to potentially overwhelm the cancer burden. [1]
- It is during this expansion phase, which can take several weeks, that careful logistical planning becomes essential. If a patient's cancer is aggressive, they may require bridging chemotherapy to manage tumor progression while waiting for their custom CAR T-cells to be grown and validated for infusion. [4] This waiting period is a critical, often stressful, bottleneck in the treatment schedule.
- Infusion: Once the cells are confirmed viable and of high quality, they are frozen, shipped back to the treatment center, thawed, and infused back into the patient, typically in a single intravenous session. [1][3]
# TCR Versus CAR
While CAR T-cell therapy targets antigens on the surface of cancer cells, another important example of T cell therapy is TCR cell therapy (T-cell receptor therapy). [5] The difference lies in what the T-cell is engineered to recognize.
In TCR therapy, T-cells are modified to express a naturally occurring T-cell receptor (TCR) that is specific to an antigen. [5] The key distinction here is that TCRs recognize antigens that have been processed inside the cell and presented on the cell surface via the Human Leukocyte Antigen (HLA) complex. [5] This capability allows TCR therapy to target proteins hidden within the cancer cell, rather than just those accessible on the exterior membrane. [5] This opens the door to treating solid tumors, which often express fewer suitable surface targets than blood cancers do. In essence, CAR T acts like a surface scanner, while TCR therapy acts like an internal scanner, provided the target is properly displayed by the HLA machinery. [5]
# Managing Risks
Although CAR T-cell therapy represents a major advancement, the infusion of highly activated, engineered immune cells carries specific risks that require specialized management. [3][5] The most common and potentially severe acute side effects are Cytokine Release Syndrome (CRS) and neurotoxicity, often referred to as ICANS (Immune Effector Cell-Associated Neurotoxicity Syndrome). [3][5]
CRS occurs when the massive activation of the infused CAR T-cells releases a large flood of inflammatory proteins, or cytokines, into the bloodstream. [3] This immune over-response can cause flu-like symptoms, high fevers, dangerously low blood pressure, and oxygen requirements, potentially affecting organ function. [3] Treating CRS often involves supportive care and the use of targeted immunosuppressive drugs like tocilizumab. [3]
Neurotoxicity symptoms range from confusion and headaches to seizures and life-threatening cerebral edema. [3] Unlike the generalized inflammatory response of CRS, neurotoxicity directly affects the central nervous system. [5] The management of these toxicities necessitates that patients receiving CAR T-cell therapy be treated in specialized hospital centers equipped with intensive care capabilities. [2][3] The targeted nature of CAR T-cells means that when the "switch" malfunctions, the resulting cascade is often swift and intense, demanding a level of acute care protocol that differs significantly from monitoring standard chemotherapy side effects.
# Therapy Impact
For patients suffering from certain refractory or relapsed B-cell malignancies—diseases that have failed to respond to multiple prior rounds of standard chemotherapy or other treatments—CAR T-cell therapy has proven to be a truly game-changing option. [6] FDA approval for CAR T-cell products has been granted for several indications, including acute lymphoblastic leukemia (ALL) in children and adults, as well as certain types of aggressive large B-cell lymphoma (DLBCL) and mantle cell lymphoma (MCL). [7][9] In cases where standard options have been exhausted, the ability of these therapies to induce deep, durable remissions in a significant fraction of patients provides immense hope. [6][9] The ongoing research is now focused on applying this powerful concept more broadly, targeting solid tumors, and developing "off-the-shelf" allogeneic products that do not require patient-specific manufacturing, which would dramatically shorten the time between diagnosis and treatment initiation. [1][5]
Related Questions
#Citations
T-cell Transfer Therapy - Immunotherapy - National Cancer Institute
CAR T-cell Therapy and Its Side Effects | American Cancer Society
CAR T-Cell Therapy: What It Is & How It Works - Cleveland Clinic
CAR T-Cell Therapy | The University of Kansas Cancer Center
Types of Cancer Treatments: T Cell Receptor (TCR) Therapy
For many blood cancer patients, CAR T-cell therapy is game-changing
FDA-approved CAR T-cell Therapies - UPMC Hillman Cancer Center
CAR T-Cell Therapy for Cancer - OHSU
CAR T-Cell Therapy | Fact Sheets - Yale Medicine

