What type of poison is a paraquat?
Paraquat is classified as a highly toxic bipyridyl herbicide. [2] Chemically known as , it is a quaternary ammonium compound that functions as a powerful, non-selective contact herbicide. [2][6] It is widely recognized not just for its agricultural effectiveness in killing weeds, but for the severe health risks associated with human exposure. [1][5] Its primary mechanism of action in plants involves disrupting photosynthesis, but in animals and humans, it causes profound cellular damage through a process of oxidative stress. [2][8]
# Herbicide Use
As a herbicide, paraquat offers rapid action, which is one reason it has been favored in certain agricultural settings globally. [6] It is used to control a wide range of grasses and broadleaf weeds and is often applied before planting or within crop cultivation systems. [3] In the United States, the Environmental Protection Agency (EPA) has registered it for use on crops such as cotton, corn, soybeans, and various fruits and vegetables, though its application is regulated. [3] It is frequently found in commercial products, one widely recognized example being Gramoxone. [7]
The speed at which it kills plant tissue—often causing visible results within hours of application—contrasts sharply with the clinical timeline for human poisoning. While a plant wilts quickly, the most devastating systemic effects in humans, particularly to the lungs, can take days or even weeks to fully manifest. [1][2]
# Cellular Damage
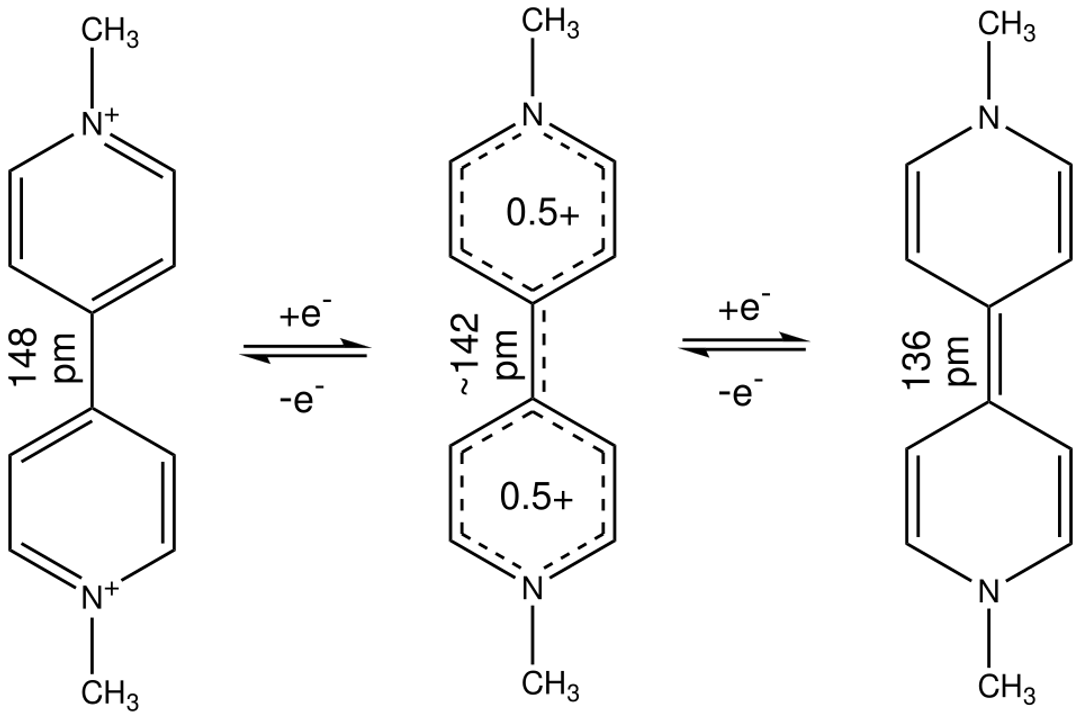
The inherent danger of paraquat lies in its unique toxicological pathway, which relies on its ability to undergo redox cycling within the body’s cells. [8] In a simplified view, paraquat readily accepts an electron, forming a highly unstable radical known as the paraquat radical. [2][8] This radical immediately reacts with molecular oxygen, regenerating the original paraquat molecule and, critically, generating highly damaging reactive oxygen species (ROS), such as superoxide radicals. [8]
This cycle repeats rapidly, consuming cellular reducing equivalents like in the process. [8] By continuously generating ROS, paraquat induces widespread oxidative stress, damaging lipids, proteins, and within the cells. [8] While this mechanism occurs throughout the body, certain tissues accumulate the compound more effectively, making them primary targets for irreversible injury. [1]
# Lung Targeting
The lung is the principal target organ in severe paraquat poisoning. [1][9] This selectivity occurs because lung cells contain high concentrations of the enzyme NADPH oxidase, which is instrumental in generating the initial superoxide radicals when paraquat is present. [8] Furthermore, the lung tissue has a relative deficiency in the protective enzyme glutathione peroxidase, which normally detoxifies these free radicals. [8]
When poisoning occurs, the damage in the lungs progresses through distinct phases. Initially, there might be alveolar damage, followed by proliferation of fibroblasts, ultimately leading to severe and often irreversible pulmonary fibrosis. [1][8] This scarring stiffens the lungs, severely impairing gas exchange and leading to respiratory failure, which is the most common cause of death in survivors of significant ingestion. [1]
# Rapid Uptake
For a poison to exert its full effect, it must be absorbed into the bloodstream, and paraquat is notoriously efficient at this process. [4] If ingested, paraquat is rapidly and nearly completely absorbed across the gastrointestinal mucosa. [4][9] This rapid uptake is one of the most concerning aspects of exposure, as it means decontamination efforts must begin almost instantaneously to be effective. [9]
Unlike some other ingested toxins, which might be partially neutralized by food or slowly absorbed, paraquat moves quickly into the systemic circulation. [4] This high efficiency of absorption means that even relatively small ingestions can lead to catastrophic outcomes. [5]
Given its rapid GI absorption and the lack of a specific, universally effective antidote, a key consideration for first responders is the volume of solution ingested relative to the patient's body mass. For instance, ingesting even small amounts (e.g., 10-15 ml of a concentrated formulation) can be fatal for an average adult, highlighting that this is not a poison where a large "dose" is required to cause catastrophe; precision in the amount swallowed matters immensely. [1][5]
# Clinical Manifestations
The signs and symptoms following exposure vary depending on the route (ingestion, dermal contact, or inhalation) and the concentration of the product involved. [6] Ingestion is the most lethal route. [1]
Initial symptoms following ingestion often begin within minutes to hours and typically involve the corrosive nature of the substance on contact tissues:
- Burning sensation in the mouth and throat. [5]
- Nausea, vomiting, and abdominal pain. [5][9]
- Potential diarrhea or bloody stools. [9]
Systemic toxicity follows this initial local irritation. Within the first few days, the patient may develop signs of multi-organ failure, including acute kidney injury and liver damage. [9] If the patient survives the initial acute phase, the focus shifts to the lungs. Pulmonary symptoms, such as shortness of breath (dyspnea), may emerge several days to weeks post-exposure, signaling the onset of fibrotic changes. [1]
Dermal exposure, especially to concentrated formulations, can cause skin irritation, blistering, and, if exposure is prolonged or over a large area, systemic absorption leading to internal organ damage. [6] Inhalation exposure, though less common outside of occupational settings, can cause irritation of the nasal passages and lungs. [6]
# Treatment Difficulties
Management of paraquat poisoning centers heavily on minimizing further absorption and providing aggressive supportive care, as there is no single, reliable antidote. [9] Immediate actions aim to sequester the poison within the gut before it enters the bloodstream. [9]
Common methods employed include administering adsorbents:
- Activated Charcoal: Used to bind the toxin in the stomach. [9]
- Fuller's Earth: A type of clay known for its high affinity for paraquat. [4][9]
When considering decontamination, the specific formulation matters. While activated charcoal is a standard binder, its effectiveness against paraquat can sometimes be reduced if the product contains surfactants intended to make it stick to leaves better; in these cases, using materials like Fuller's Earth, which has a very high adsorptive capacity specifically for this molecule, becomes the preferred intervention according to some clinical guidelines. [4][9]
If ingestion was very recent (within the first hour), gastric lavage might be considered, though this carries its own risks. [9] Once the poison is absorbed, treatment becomes largely supportive, focusing on maintaining kidney and lung function and managing the body’s overwhelming oxidative stress, often requiring intensive care monitoring. [1][9] The prognosis remains poor for patients who ingest large amounts because the resulting lung fibrosis is often progressive and irreversible. [1]
# Global Regulation
The extreme toxicity and lack of effective treatment have led to significant regulatory action worldwide. [2] In many regions, including the entire European Union, paraquat is completely banned due to the high incidence of fatal self-poisoning cases. [2]
However, its continued use in countries like the United States highlights a regulatory divergence based on perceived risk management and agricultural necessity. [3] Where it is still permitted, strict safety measures, personal protective equipment requirements, and restricted-entry intervals are mandated by regulatory bodies to minimize occupational and environmental exposure. [3][6] This global patchwork means that what is available on the shelf in one agricultural zone is strictly prohibited just across an international border, affecting how the chemical is perceived and handled globally. [2]
Related Questions
#Citations
Paraquat Poisoning: Symptoms & Treatment - Cleveland Clinic
Paraquat - Wikipedia
Paraquat Dichloride | US EPA
Paraquat Herbicide Information
Paraquat poisoning: MedlinePlus Medical Encyclopedia
Paraquat Fact Sheet - National Pesticide Information Center
What Products Contain Paraquat? | ELG Law
Paraquat: The Poison Potion - PMC - PubMed Central - NIH
Paraquat and diquat poisoning - UpToDate


